[ad_1]
Scientists at the University of Warwick have created a new way to view proteins that are inside human cells.
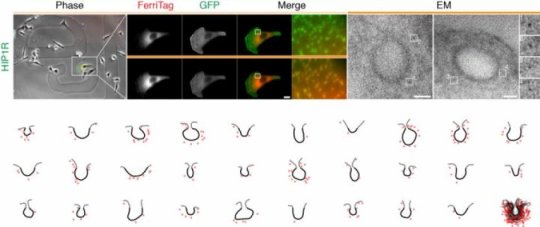
Using Ferritin, a large protein shell that our cells use to store iron, the researchers have found a method they have called FerriTag that allows an electron microscope (EM) to view proteins precisely unlike current methods.
The method allowed the scientists to enable the cell to make the tag itself avoiding damage caused by placing it from the outside of the cell. Their paper FerriTag is a new genetically-encoded inducible tag for correlative light-electron microscopy is published in the journal Nature Communications.
The team set out to precisely localise a protein found in clathrin-coated pits. These are 100 nm wide entry points used by viruses to invade cells and infect them. Using FerriTag, the team were able to see where the protein is found in the pit and on the inside face of the cell’s surface.
The team was led by Dr Stephen Royle Associate Professor and Senior Cancer Research UK Fellow at Warwick Medical School. He said: “Proteins do almost all of the jobs in cells that scientists want to study. We can learn a lot about how proteins work by simply watching them down the microscope. But we need to know their precise location.”
Although light microscopy can be used to view proteins move around the resolution is low, so seeing a protein’s precise location is impossible. This can be overcome by using electron microscopy which gives a higher resolution.
To allow proteins to be viewed by both microscopes and correlate them, the research team developed a method of tagging the proteins so that they can be seen by both types of equipment.
Tagging is widely used and several tags are available however they have established drawbacks; some are not precise enough, or they don’t work on single proteins. To overcome this Dr Royle’s lab created a new tag and fused it with a fluorescent protein.
Dr Royle’s team named the new technique FerriTag because it is based on ferritin which can be viewed by an electron microscope because iron scatters electrons.
Dr Royle said his lab had to defeat another obstacle: “When Ferritin is fused to a protein, we end up with a mush. So, we altered Ferritin so that it could be attached to the protein of interest by using a drug.
“This meant that we could put the FerriTag onto the protein we want to image in a few seconds.
“The cool thing about FerriTag is that it is genetically encoded. That means that we get the cell to make the tag itself and we don’t have to put it in from outside which would damage the cell.”
Story Source:
Materials provided by University of Warwick. Note: Content may be edited for style and length.
[ad_2]