[ad_1]
With age, our tissues lose their function and capacity to regenerate after being damaged. A study published today in Cell by scientists at the Institute for Research in Biomedicine (IRB Barcelona) and the Centro Nacional de Análisis Genómico of the Center for Genomic Regulation (CNAG-CRG) explains how dermal fibroblasts age.
The main conclusion drawn is that these fibroblasts lose their cell identity, as if they had “forgotten” what they are, and consequently their activity is altered, thus affecting tissue. The study reveals the cellular and molecular pathways affected by ageing and proposes that they could be manipulated to delay or even reverse the skin ageing process.
The skin
Dermal fibroblasts are key for the production of collagen and other proteins that make up the dermis and that preserve the skin’s function as a barrier. The activity of these cells is also crucial for the repair of skin damage. As we age, the dermis loses its capacity to produce collagen, and consequently its capacity to repair wounds is also significantly impaired.
“The elderly face many problems in this regard because their skin does not heal properly and its barrier properties are decreased, thus increasing the risk of skin infections and systemic infections,” explains Salvador Aznar Benitah, ICREA researcher at IRB Barcelona and leader of the study. “The notion that the loss of cell identity is one of the underlying causes of ageing is interesting and one that we believe hasn’t been considered before,” he concludes.
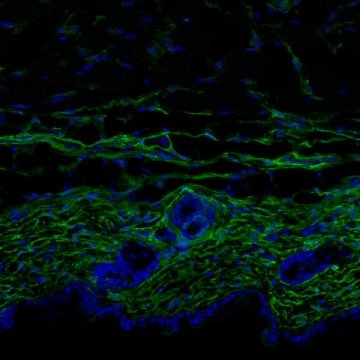
Holger Heyn, team leader at the CNAG-CRG and co-leader of the study, explains that “cutting-edge technologies allow molecular analysis of individual cells. In this study, we have applied advanced techniques to obtain high resolution images of fibroblasts as they age.”
First-authored by IRB Barcelona PhD student Marion Salzer, the study demonstrates that, during ageing, skin fibroblasts start to acquire many traits that are characteristic of adipocytes (fat cells). “This leads to them losing their cell identify and they also stop producing and secreting collagen like they should,” explains Salzer.
The single-cell analysis confirmed the loss of fibroblast identity in aged animals. Using sophisticated computational tools, the scientists observed that aged fibroblasts show a less defined molecular conformation compared to young fibroblasts and that “they resemble the undefined cell states observed in newborn animals,” says Heyn.
Although being basic research, Aznar Benitah comments that “this new knowledge might not only have cosmetic applications, for example anti-aging skin treatment, but more importantly, also therapeutic applications aiming to help aged skin to form scar tissue faster and more efficiently after being damaged or after an operation.”
Story Source:
Materials provided by Center for Genomic Regulation. Note: Content may be edited for style and length.
[ad_2]