[ad_1]
A collaborative research group led by Kumamoto University has developed a new control system for regulating the morphology and orientation of cells that constitute animal tissues.
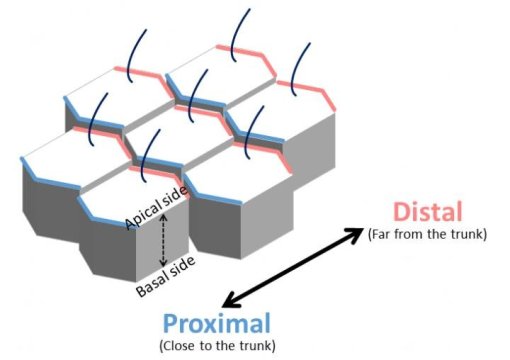
Cells that make up animal tissues must have proper form and an ordered arrangement. This behavior is determined by cell polarity where the opposite ends of a cell, such as up/down or front/back, have different properties. For example, the hair that grows on the surface of Drosophila (fruit fly) wings is aligned in a particular direction because it grows only from the outer part of each cell that forms the surface of the wing. This is called planar cell polarity (PCP).
Tissues will not form correctly if cell polarity formation is impaired, and malformation can lead to the development of various diseases. Furthermore, maintaining cell polarity is necessary for preserving tissue homeostasis. Failure of this mechanism can lead to carcinogenesis and malignant cancer.
Since the formation of cell polarity involves a change in cell morphology, it is necessary to change the dynamics of the structure that determines the morphology of the cell, the cytoskeleton. It is known that changes in the cytoskeleton dynamics are induced by intracellular messengers (signaling molecules) and it has been suggested that the cytoskeleton not only receives a signal at the time of kinetic changes, but may also produce feedback through some form of signal transduction. However, the mechanism linking cytoskeletal dynamics and the signaling mechanisms has not yet been clarified.
Researchers from Kumamoto University focused on the fact that the Wnt5a signaling pathway controls the front-rear polarity (i.e., the cell shape during movement) of cells derived from human cervical cancer. They found a microtubule-associated protein, Map7/7D1, which links the Wnt5a signaling pathway and microtubule dynamics by associating with Dishevelled (Dsh, or Dvl in mammals) protein, a transmitter in the Wnt5a signaling pathway.
The Wnt5a signaling pathway is already known to be essential for PCP formation in epithelial cells. Researchers therefore examined the function of Map7/7D1 during PCP formation using Drosophila wing and mouse oviduct epithelial tissues. They found that the protein equivalents to Map7/7D1 and Dvl in Drosophila, Ens and Dsh, bind together, and that Ens controls the localization of Dsh to the distal side during PCP formation.
Furthermore, Map7/7D1/Ens showed planar-polarized distribution at the ovary/proximal side in epithelial cells of mouse oviducts and Drosophila pupal wings, respectively. Since the behavior and properties in cells (such as localization patterns and binding with Dvl/Dsh) are conserved, the researchers suggest that the functions of Map7/7D1/Ens are conserved and inherited similarly in many different species during PCP formation. They believe they have discovered a new control system for regulating the morphology and orientation of tissue cells.
This research was posted online in the journal “EMBO Reports” on 7 June 2018.
Story Source:
Materials provided by Kumamoto University. Note: Content may be edited for style and length.
[ad_2]