[ad_1]
During brain development, billions of neuron nerve cells must find their accurate pathway in the brain in order to form trillions of neuronal circuits enabling us to enjoy cognitive, sensory and emotional wellbeing. To achieve this remarkable precision, migrating neurons use special protein receptors that sense the environment around them and guide the way so these neurons and their long extensions stay on the right path and avoid faulty turns. Rare defects in these neuronal guidance proteins can result in severe neurological conditions such as ataxia and epilepsy.
In a new study published in Cell, Bar-Ilan University researchers and collaborators report on their discovery of the intricate molecular mechanism that allows the guidance receptor “Robo” to react to signals in its environment, while avoiding premature activity that can lead to harmful outcomes.
One of the most important protein signaling systems that controls neuronal guidance consists of the cell surface receptor “Robo” and its cognate external guidance cue “Slit.” The deficit of either of these proteins results in defects in brain structure and function. For example, their absence compromises the brain’s ability to form the correct connection across the corpus callosum, which is the region where neuronal extensions from the two brain hemispheres cross paths to innervate opposite sides of the body, a fundamental attribute of bilateral creatures.
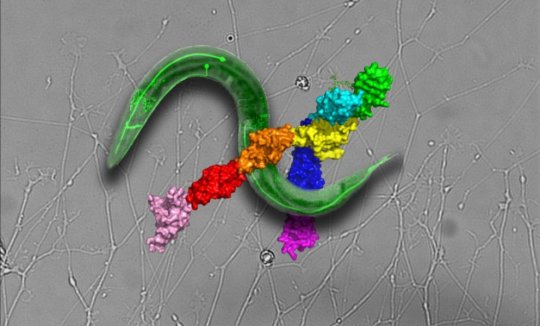
“Slit and Robo are highly conserved throughout evolution, and can be identified in virtually all animals with a nervous system, from a one-millimeter-long nematode all the way to humans, an aspect that highlights their importance,” explains Prof. Yarden Opatowsky, Director of the Laboratory of Structural Biology in the Mina and Everard Goodman Faculty of Life Sciences at Bar-Ilan University, Israel. Using X-ray crystallography, Opatowsky, graduate student Reut Barak, and their colleagues and collaborators determined several atomic structures of Robo. “It took us six years to get the desired structure, but it was just the first phase of our research,” says Opatowsky. The structures indicated very nicely how two Robo receptors form dimers and how their dimerization interfaces can be blocked.” This led lab manager Dr. Julia Guez-Haddad and graduate student Galit Yom-Tov to embark on a series of biochemical and functional experiments to follow up on the structure-driven hypotheses.
A comprehensive model for Robo activation
Since their discovery about two decades ago, there has been a lot of progress towards understanding Slit-Robo function and the developmental responses that they trigger in the brain, as well as outside the central nervous system. However, scientists still have an incomplete understanding of how Slit activates Robo and what keeps Robo from getting activated in the absence of Slit. Following up on the new crystallographic observations, Opatowsky’s team developed a new biochemical assay that allowed them to monitor Robo dimerization when expressed in cultured cells. In this way, they corroborated the structural model for Robo’s self-control over its oligomeric state and also showed that the same mechanism actually exists in other closely related receptors. But does the new model hold true in an actual living biological system? Opatowsky explains: “Here we took a reductionist approach by examining the fate of one specific neuron type. In the course of evolution, Robo and Slit were duplicated several times, a process that resulted with humans and mice having four Robo genes and three Slits, where each copy has its own special attributes, but also maintains considerable redundant functions. We figured that conducting our experiments in creatures with such a high level of Robo and Slit redundancy may turn out to be like playing genetic ‘whack-a-mole’, where knocking out one Robo or Slit copy will be compensated by the upregulation of another copy. Luckily for us, the tiny C. elegans nematode has only one Robo and one Slit and was previously characterized for Slit and Robo function, which makes it ideal for our purposes. The evolutionary distance between humans and C. elegans is about 500 million years, and yet, all the experiments that we conducted point toward a similar Robo molecular mechanism.”
Robo is an attractive drug target
When Robo and Slit are not expressed properly they are involved in the formation and progression of various developmental and chronic conditions, such as age-related macular degeneration, loss of bone mass, and kidney diseases. The involvement of Slit and Robo in cancer is of particular interest to researchers. Normally our cells rely on external signals for their growth, differentiation, migration, and eventual elimination. These external signals activate cellular receptors that relay the correct instructions into the cell. In cancer, some receptors are “hijacked” to drive tumor formation, progression and metastasis. In personalized cancer therapy based on the genetic profile of the patient’s tumor, drugs are used to block these rogue receptors, thereby depriving cancer cells with vital signaling instructions and directing them toward destruction. “Slit and Robo are aberrantly expressed in cancer and targeting them is long considered a promising therapeutic approach for incurable subtypes of pancreatic, skin and breast cancer,” says Opatowsky. “However, there are currently no Robo directed drugs, and we hypothesized that it is due to an insufficient structural and mechanistic understanding of Robo activation and signaling. Our discoveries provide, for the first time, the information necessary to design effective drugs targeting Robo receptors. In particular, the crystal structures revealed molecular sites on the surface of Robo that when targeted by designed drugs will allow us to manipulate Robo activation and inhibition in patients, thus providing new possibilities for treatment in the war on cancer.”
Story Source:
Materials provided by Bar-Ilan University. Note: Content may be edited for style and length.
[ad_2]